 |
| A confiscated moonshine
still shows the basic components of a distillation apparatus |
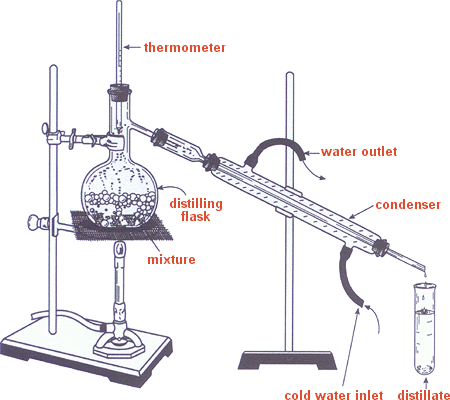
- Set up a distillation apparatus as shown in above. (See Mr Arrow after school) Check the following
details:
- The top of the thermometer bulb should be just below the side arm of the distilling flask, ensuring that the entire thermometer bulb will be bathed in the rising vapor as the liquid mixture is heated.
- The side arm should extend beyond the cork in the top end of the condenser, and the condenser tip should extend beyond the cork in the adapter. The distillate cannot pick up impurities from the corks when set up this way.
- Both the distilling flask and the condenser must be firmly clamped in place.
- To prevent having a closed system, the lower end of the adapter should not be connected to the receiver by a cork. This arrangement prevents pressure from building up and causing an accident.
- Begin circulating water from the lower end of the condenser prior to heating the distilling flask.
- Add two or three boiling chips (small marble chips or porcelain chips) to the distilling flask to prevent superheating and bumping.
- Have your teacher check the setup before applying heat to the flask.
- Label and number four large test tubes (20- x 150-mm) for collecting fractions. Place Tube 1 under the condenser outlet.
- Remove the thermometer with its cork. By means of a long-stem glass funnel, add about 20-30 ml 2-propanol-water mixture to the distilling flask.
- Gently heat the flask until the liquid begins to boil. Take temperature readings every half-minute and record these along with the time. Continue heating just enough to keep the liquid boiling while continuing to take temperature readings. Continue boiling the liquid until the temperature remains the same for about 3-4 minutes.
- Graphing the data from Step 4 provides the information needed to decide about the temperature range to collect separate fractions. Do the graphs before continuing. Repeat the procedure (Steps 3-4) but this time use the data collected in Step 4 to collect separate fractions of distillate in tubes #2, #3, and #4. You should be able to detect the presence of at least three separate fractions, the last one being essentially pure water.
- Cautiously smell each fraction and record your observations. Test the flammability of each fraction by dipping a small piece of paper in each and trying to ignite the wet paper with a match. Use a balance and graduated cylinder to measure the density of each fraction. Test the solubility of sugar in a small amount (about 1 ml) of each fraction. Record all observations.
- Thoroughly wash your hands before leaving the laboratory.
- Can you tell just by looking that the clear liquid is a mixture?
- Does the paper dipped in the liquid burn?
- What is the density of the mixture prior to distillation?
- What is the density of each fraction?
- Does sugar dissolve in each fraction?
- What do you conclude about the composition of each fraction?
- On the basis of the tests performed on the original solution and each fraction, what can you conclude about the composition of each fraction?
- What do you think would be the result if you redistilled each fraction into three fractions of equal volume?
- What are the differences in properties that may be used to separate the components of a mixture?
- What purpose is served in separating the components of mixtures?
- How do components of a mixture affect the properties of a mixture?
- How does a mixture differ from a compound?
- How does a homogeneous mixture differ from a heterogeneous mixture?
- What are some household products that are the result of separation techniques?
For more information, at other Web sites...
- The Law is Coming from That-a-Way — Article recounting the
life and times of North Carolina Alcoholic Beverage Control Officer Arthur
R. Currin, featuring information on bootleg liquor distillation in American
history and descriptions and photographs of apparatus used by old-time
bootleggers, written by Cynthia M. Currin.
Antibiotics in Action Directory | Site Map | Pharmaceutical Achievers Home